-
- Posts: 3
- Joined: Sat Sep 28, 2013 3:02 am
I'm utilizing peptide-level size exclusion chromatography to separate tryptic peptides on an AKTA Fast Protein Liquid Chromatography system with a Superdex Peptide PC 3.2/30 gel filtration column (ideal separation from 100-7000 Da). We utilize an isocratic elution with a mobile phase consisting of 30% Acetonitrile and 0.1% TFA at 50 μL/min (bed volume 2.4 mL). In order to calibrate our separation, we use insulin (5.7 kDa) and angiotensin II as standards. While running standard separations today, I noticed a few ghost peaks and substantial baseline drift.
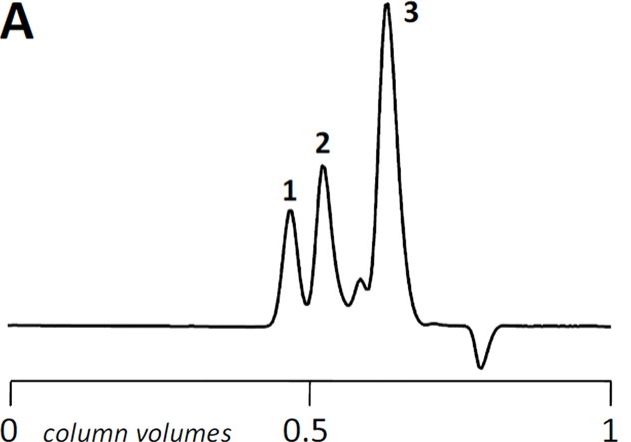
Normally, our standard run has the following UV chromatogram at 215 nm:
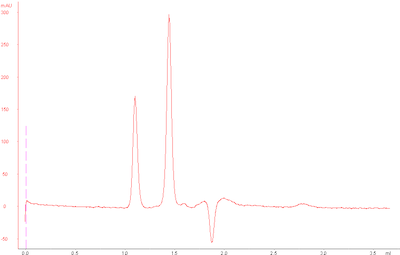
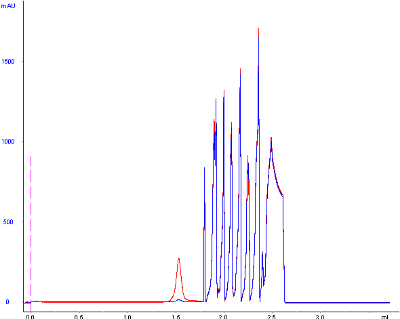
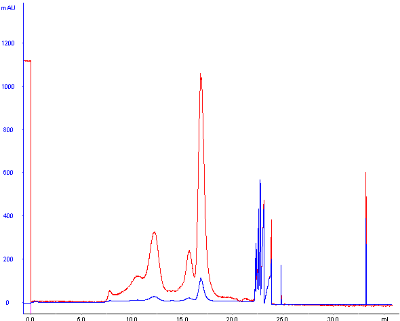
However, my standard runs today looked as follows:
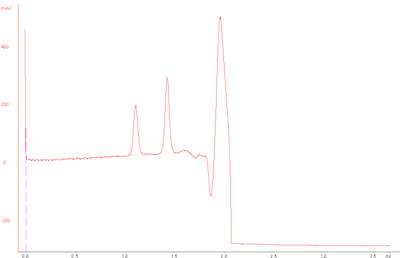
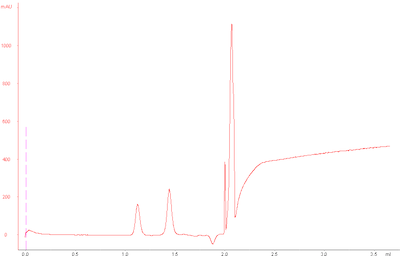
The base peak drift seems to occur after small molecules would typically elute , since glycine (75 Da) elutes at around 2.0 mL.
http://eclub.biomart.cn/sites/eclub.bio ... 3.2-30.pdf
I'm confused as to the possible explanations of these ghost peaks and the substantial baseline drift, as the peaks should not be occur from samples left on the column after a previous run (small molecules elute well within the separation, and these standards are all in high-purity and well above that molecular weight range), I include an extra 2 CV wash between sample runs to fully elute any and all molecules injected onto the column. Furthermore, since the separation is isocratic, the baseline should not drift dramatically for any reason.
Could this potentially be due to an old UV lamp? I doubt it, but I've eliminated many other possibilities and our UV lamp is quite old at this stage.
EDIT:
One thing I am noticing is that these ghost peaks and baseline drifts are identical at 280 nm and 215 nm, suggesting that the solvent or any real elution itself is not to blame. Am I right in suggesting that the UV lamp is the predominant cause?