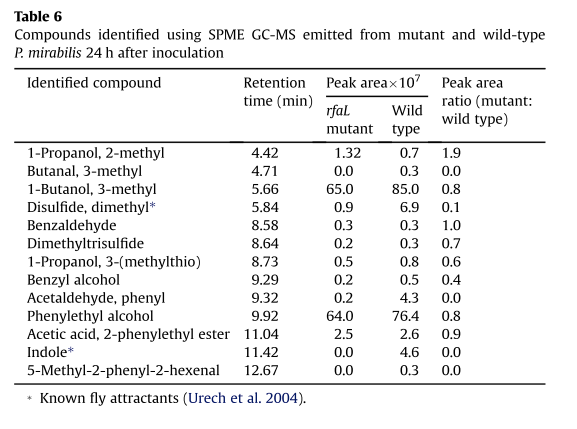
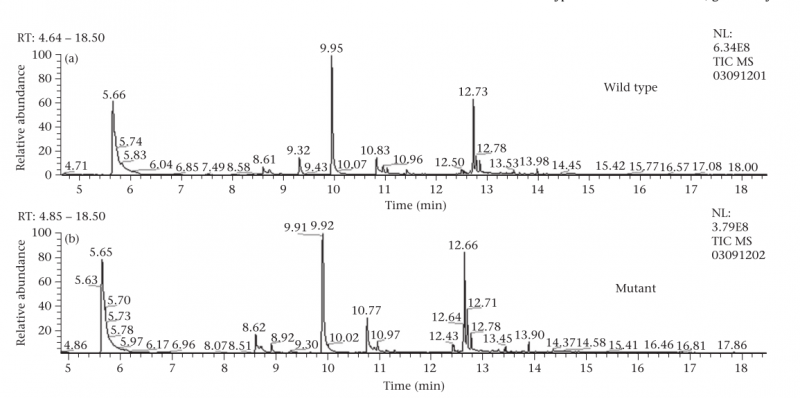
-
- Posts: 11
- Joined: Thu Apr 18, 2013 3:15 pm
I'm developing a protocol to analyse headspace from a gram negative bacteria in log phase growth.
I've been asked to use an internal standard to reduce the variability and the analyst who runs the GC-MS and one or two others suggested Decane. I'm analysing 5ul bacteria on 3ml LB agar in a 20ml autosampler vial, adding the Decane around 10 minutes before the bacteria is added. We found that 10ul pure Decane almost saturates the column, so now my analyst thinks I should use just 0.1ul of standard. I believe that's pretty much impossible given the inaccuracy of pipetting such a small amount. I had the idea of diluting, in Hexane, but perhaps this will not help if I can only put 0.1ul of anything in there. My analyst thinks diluting will make no difference, my lab instructor thinks it will be fine and I'm stuck in the middle since I have very limited knowledge of this and almost no training in this method. I've done a bit of digging around, but I can't seem to find anything about using internal standards for this particular system.
Also, I noticed that one of the fibres has some part of it missing...ie I can see the wire it's attached to. Does this spell the end for that particular fibre? We always pierce the septum with the fibre retracted so it doesn't bend.
If anyone has as experience with this or suggestions, I'm all ears. Also, if there's any specific details which would help you answer my query then let me know.
Thanks!
Lue