-
- Posts: 2
- Joined: Thu Mar 03, 2011 1:55 pm
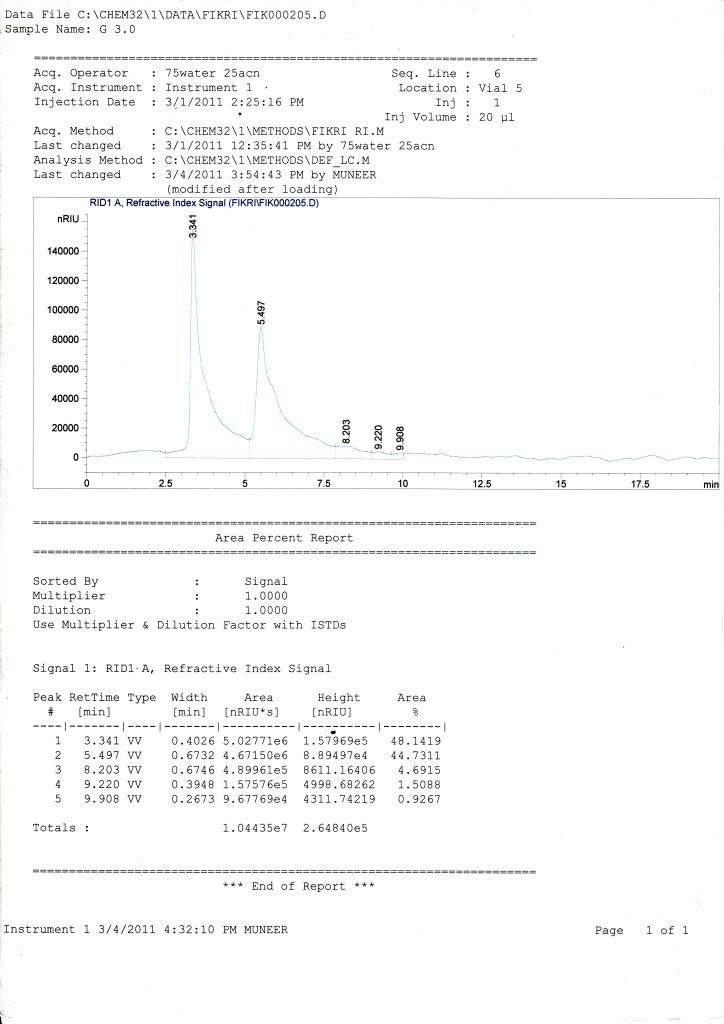
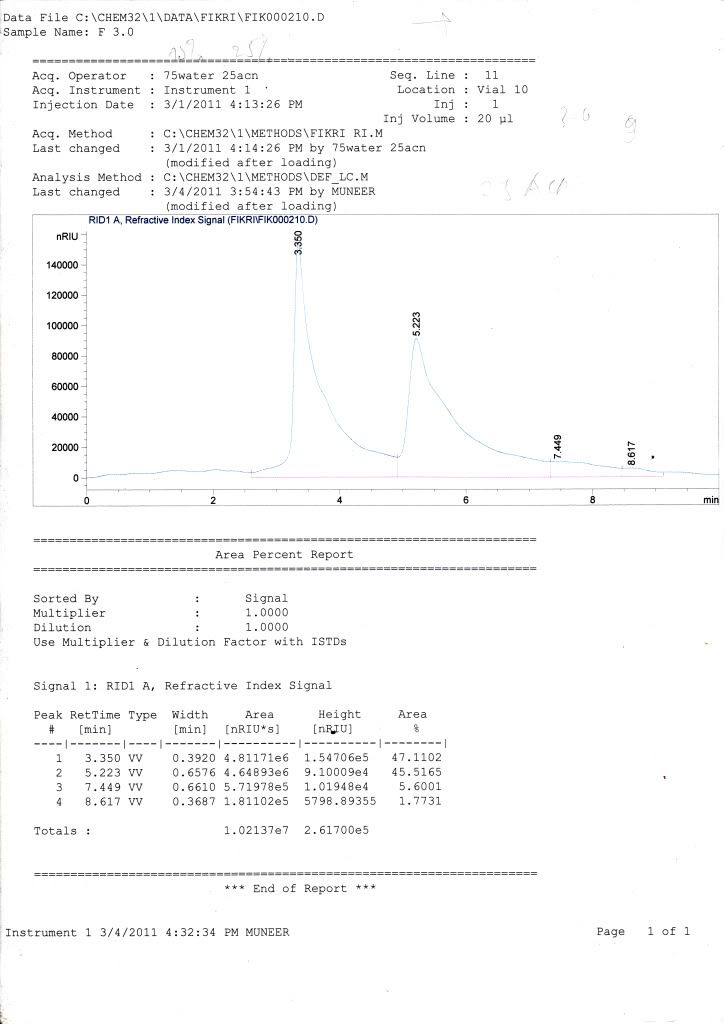
sample = glucose + fructose in phosphate buffer (pH 6-8)
mobile phase = 75% ACN + 25% water
column = kromasil
flow rate = 1 mL/min
temperature = 40 C
for my standard curve, retention time for glucose is 5.2 while for fructose is 5.1 ( i dont know whether the retention time is logic or not)
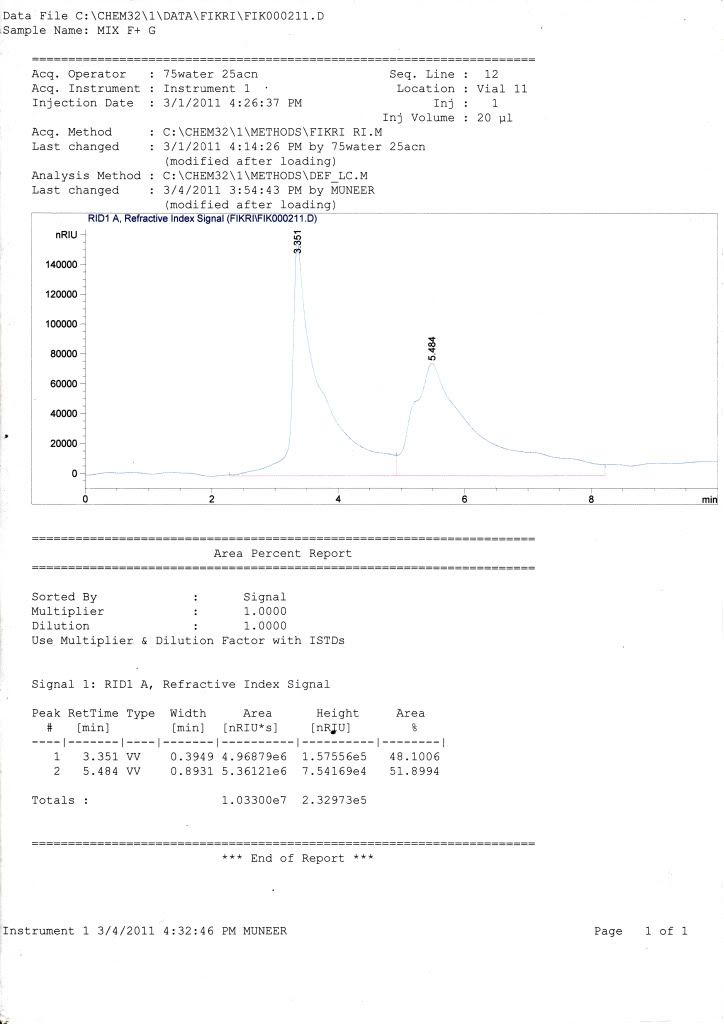
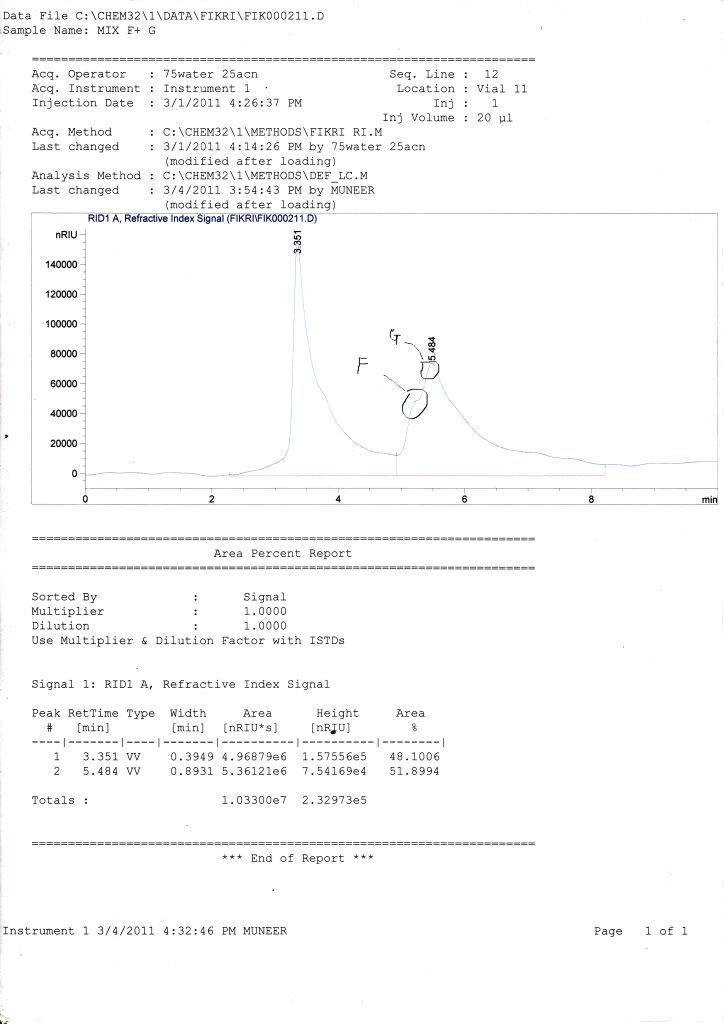
for my samples, all graph only show 1 peak of retention time but have two curves (small one for fructose while the large one for glucose) where HPLC cant fully separate it (maybe because the retention time difference is too small or maybe effect from the phosphate buffer in my sample.)
i already tried using 60% ACN + 40% water, 100% water, and flow rate around 0.5-1 mL/min but the result still same with the previous one and really difficult for me to identify area of fructose and glucose. can anybody help me to find the solution?